Class 12 Chemistry CBSE Chemical Kinetics Mostlikely QuestionBank
Here we provide Class 12 chemistry important notes,board questions and predicted questions with Answers for chapter Chemical Kinetics. These important notes,board questions and predicted questions are based on CBSE board curriculum and correspond to the most recent Class 12 chemistry syllabus. By practising these Class 12 materials, students will be able to quickly review all of the ideas covered in the chapter and prepare for the Class 12 Board examinations as well as other entrance exams such as NEET and JEE.
class 12 CBSE Chemical-Kinetics Mostlikely-QuestionBank
Chemical-Kinetics Mostlikely-QuestionBank
Q1
For a reaction:
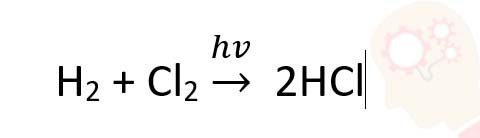
(i) write the order and molecularity of the reaction
(ii) write the unit of k
solutions
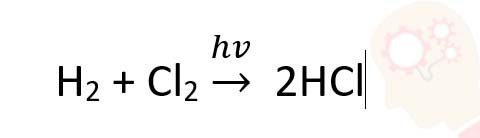
(i) write the order and molecularity of the reaction
(ii) write the unit of k
solutions

Q2
A first order reaction takes 30 minutes for 50% completion calculate the time required for 90%
completion of this reaction.(log 2 = 0.3010)
solutions

solutions


Q3
The rate of reaction becomes four times when the temperature changes from 293 K to 313 K. calculate
the energy of the activation (Ea) of the reaction assuming that it does not change with
temperature.
solutions
solutions

Q4
Calculate the half life period of a first order reaction with the rate constant is 5
years-1
solutions
solutions


Reach Us
SERVICES
- ACADEMIC
- ON-LINE PREPARATION
- FOUNDATION & CRASH COURSES
CONTACT
B-54, Krishna Bhawan, Parag Narain Road,
Near Butler Palace Colony Lucknow
Contact:+918081967119

Add a comment